CTIX
01/21/2012
CTIX (neutral, speculative buy if funds start accumulation), 10/02/13 - Strong Sell
03/04/16 CTIX status scam was changed to "good biotech" - recommendation BUY and HOLD
The Styx (the variant spelling Stix) - Ancient Greek: Στύξ, also meaning "hate" and "detestation") (adjectival form: Stygian, /ˈstɪdʒiən/) is a river in Greek mythology that formed the boundary between Earth and the Underworld (often called Hades which is also the name of this domain's ruler). It circles the Underworld nine times. The rivers Styx, Phlegethon, Acheron, and Cocytus all converge at the center of the underworld on a great marsh, which is also sometimes called the Styx.
CELLCEUTIX CORP
Cellceutix Corporation, a development stage biopharmaceutical company, engages in the discovery and development of small molecule drugs to treat unmet medical conditions. The company's lead product candidate includes Kevetrin, which is in pre-clinical development stage primarily for the treatment of head and neck cancers. It is also developing various other product candidates for the treatment of psoriasis, rheumatoid arthritis, osteo-arthritis/asthma, hypertensive emergency, and autism, as well as neurological disorders, such as multiple sclerosis, Lou Gehrig disease, and/or Parkinson's disease. Cellceutix Corporation has a collaborative agreement with Pioneer Valley Life Sciences Institute to investigate Kevetrin's role in antitumor activity in the context of age-related mitochondrial dysfunction. The company was founded in 2007 and is headquartered in Beverly, Massachusetts.
KevetrinCancer Preclinical
KM 277 Arthritis Preclinical
KM 278 Arthritis/Asthma Preclinical
KM 133 Psoriasis Preclinical
KM 3174 Cancer Early R&D
KM 362 MS/ALS/Park Early R&D
KM 732 Hypertensive emergency Early R&D
KM 391 Autism Preclinical
When biotech investor see introduction like this he should say just: WOW! This microcap is a future biotech gem!
But sometimes small biotechs are just founded for only one purpose: to fool biotech investors and may be some funds. The founders sell shares and become rich. We (investors) have losses and usually say to yourself: Why I was so stupid?
I always wanted to catch some small biotech at the beginning i.e. when they start to fool investors. Especially to fool institutions and funds...
May be CTIX (or Stix) is a good catch.
Details
Index Membership: N/A
Sector: Healthcare
Industry: Biotechnology
Full Time Employees: 2
Cellceutix announced that research on KevetrinTM, the Company's flagship compound against cancers, has demonstrated the potential for a major breakthrough in cancer research by exhibiting an activation of p53in both wild and mutant types of p53. p53, often referred to as the "Guardian Angel Gene" or the "Guardian Angel of the Human Genome" due its crucial role in controlling cell mutations, is a tumor suppressor protein that is encoded by the TP53 gene in humans and has been widely regarded as possibly holding a key to the future of cancer therapies.
Cellceutix was formed in May of 2007 by Dr. Krishna Menon and Leo Ehrlich. The organization will develop the innovations of Dr. Menon and his team in the laboratory. The company owns intellectual property stemming from the inventions of Dr. Krishna Menon, whose background consists of over 35 years of experience working as team leader in drug development for top pharmaceutical companies and academic institutions. The Company also owns intellectual property developed by several prominent scientists and researchers.
So, what is KevetrinTM?
Kevetrin™ - Our lead compound
has demonstrated the potential for a major breakthrough in cancer research by exhibiting an activation of p53. p53, often referred to as the "Guardian Angel Gene" or the "Guardian Angel of the Human Genome" due its crucial role in controlling cell mutations, is a tumor suppressor protein that is encoded by the TP53 gene in humans and has been widely regarded as possibly holding a key to the future of cancer therapies. p53 has been shown to play critical roles in the homeostatic health of the human body by activating proteins required to repair DNA and plays a major role in the life cycle of cells by inducing cell cycle arrest and apoptosis to maintain cellular and genetic stability.
In more than 50 percent of all human carcinomas, p53 is limited in its anti-tumor activities by mutations in the protein itself. Currently, there are greater than 10 million people with tumors that contain inactivated p53, while a similar number have tumors in which the p53 pathway is partially abrogated by inactivation of other signaling components. This has left cancer researchers with the grand challenge of searching for therapies that could restore the protein’s protective function, which Kevetrin appears to be doing the majority of the time.
Excellent results in animal model experiments in drug-resistant cancers.
Kevetrin, our lead product candidate, is a small molecule compound proprietary to the Company. Its structure is distinct from other anti-cancer agents currently on the market. Kevetrin was discovered by the Company's founder, Dr. Krishna Menon, and has been studied extensively (in vitro and in vivo) demonstrating potent anti-cancer activity against various cancer cell lines. Kevetrin's recent success in a series of animal model experiments with drug-resistant cancer cell lines, has galvanized the Company to focus on Kevetrin's development potential in this area. While Kevetrin's primary mechanism of action was thought to be AKT inhibition, more recent research leads us to look elsewhere.
Small molecule drug that is structurally different from anti-cancer agents currently on the market
Well, it's true: this molecule is a unique...
In vitro cytotoxicity is less than paclitaxel but tumor growth reduction is greater in models of drug resistant lung, breast and colon cancer
Demonstrated success in more than 5,000 small animal tests, achieving significant delays in tumor growth compared to controls in breast, prostate and colon cancer tumors.
In animal tests of a head and neck cancer cell line, delay in tumor growth was significantly increased by 14 days with Kevetrin alone (about the same as with radiation alone) but when Kevetrin was administered in conjunction with radiation, tumor growth delay increased by 36 days, more than two-fold compared to controls
In small animal tests, Kevetrin was well tolerated.
Summary
Kevetrin is effective in mouse models of human lung cancer : A549
• Kevetrin (200 mg/kg IVIP x 3 doses)
◦ 33% to 111% tumor growth delay compared to controls
◦ 33% to 100% tumor growth delay compared to paclitaxel (22 mg/kg IV x 4 doses)
• Only 3% to 4% decrease in animal weight
So, Dr. Menon says that his Keyvorit - sorry for mistake - Kevetrin activates p53 in both wild and mutant types of p53 ("Guardian Angel of the Human Genome")
But why he forgot to mention this mechanism in his patent application?
Wiki:
"p53 pathway: In a normal cell p53 is inactivated by its negative regulator, mdm2. Upon DNA damage or other stresses, various pathways will lead to the dissociation of the p53 and mdm2 complex. Once activated, p53 will induce a cell cycle arrest to allow either repair and survival of the cell or apoptosis to discard the damaged cell. How p53 makes this choice is currently unknown.
Activated p53 binds DNA and activates expression of several genes including WAF1/CIP1 encoding for p21. p21 (WAF1) binds to the G1-S/CDK (CDK2) and S/CDK complexes (molecules important for the G1/S transition in the cell cycle) inhibiting their activity."
A mutant p53 will no longer bind DNA in an effective way, and, as a consequence, the p21 protein will not be available to act as the "stop signal" for cell division. Thus, cells will divide uncontrollably, and form tumors.[26]
The critical event leading to the activation of p53 is the phosphorylation of its N-terminal domain. The N-terminal transcriptional activation domain contains a large number of phosphorylation sites and can be considered as the primary target for protein kinases transducing stress signals.
If the TP53 gene is damaged, tumor suppression is severely reduced. People who inherit only one functional copy of the TP53 gene will most likely develop tumors in early adulthood, a disease known as Li-Fraumeni syndrome. The TP53 gene can also be damaged in cells by mutagens (chemicals, radiation, or viruses), increasing the likelihood that the cell will begin decontrolled division. More than 50 percent of human tumors contain a mutation or deletion of the TP53 gene.[33] Increasing the amount of p53, which may initially seem a good way to treat tumors or prevent them from spreading, is in actuality not a usable method of treatment, since it can cause premature aging.[34]
It seems like that RCM, PhD, VMD - Dr. Krishna Menon discovered a cancer cure. And his magic drug activates even mutant p53 (may be even p53 with N-terminal domain deletion). What about people that have only one functional copy of the TP53 gene? Will Kevetrin help them?
CTIX Market Cap ($)54.6M
Shares Out. 92.5M
Major Direct Holders
MENON KRISHNA 32,048,286 Dec 10, 2007
EHRLICH LEO 7,745,002 Dec 10, 2007
So, these 2 founders are have >40% of company. If some big pharma buy it now ($1 per share) they will be millionaires.
Questions is why nobody buy this small biotech?
One can say that nobody buys such small biotech...
But recently Merck acquires the Beverly, Mass.-based company’s “SmartInsulin” technology, a self-regulating, once-a-day injectable formula that reduces the incidence of hypoglycemia and handles both meal and non-meal times of the day.
Specifically, the smart insulin tech regulates itself by responding to the plasma concentration of a molecular indicator — that is, within a specific range of glucose concentration.
The product is currently in pre-clinical, or non-human, testing. It was originally developed by Todd Zion at the Massachusetts Institute of Technology.
“If this investigational technology is ultimately approved for use with patients, it could provide an important new therapy for the treatment of diabetes,” Merck Research diabetes and obesity chief Nancy Thornberry said in a statement.
Merck will acquire all outstanding stock of SmartCells, with SmartCells shareholders receiving both cash payment and eligibility to receive clinical development and regulatory milestones for products, “for potential aggregate payments in excess of $500 million.”
$500M just "for pre-clinical, or non-human, testing"?
Why not pay just $100M for drug that delay 33% to 111% tumor growth in mice?
Recent news from CTIX:
Cellceutix Anti-Cancer Drug Shown to Regulate HDAC2, a Major Therapeutic Target for Treatment of a Broad Range of Cancers
6:30a ET January 17, 2012 (Market Wire) Cellceutix Corporation (OTCBB: CTIX) ("the Company"), a biopharmaceutical company focused on discovering and developing small molecule drugs to treat unmet medical conditions, including drug resistant cancers, announced that recent research has shown that its flagship anticancer compound Kevetrin(TM) has potent anticancer activity in a wide range of tumor types by targeting histone deacetylase (HDAC).
Cellceutix consultant Dr. Ashok Kumor recently made the discovery during the Company's research to further define Kevetrin's Mechanism of Action (MOA). Prior investigation of the MOA has shown that Kevetrin(TM) activates both transcription-dependent and transcription-independent pathways to induce apoptosis in tumor cells through activation of p53, the "Guardian Angel of the Human Genome," and potentially the retinoblastoma protein, or Rb, pathway. Damaged or mutated p53 or Rb is exhibited in nearly 100 percent of all cancers, regardless of origin. The HDAC2 connection further delineates the MOA behind Kevetrin's powerful antitumor activity that has been demonstrated against multiple cancers.
Inhibition or downregulation of HDAC reduces tumor growth and survival through cell cycle arrest, blocking of angiogenesis, an increase of antigenicity of tumor cells and induction of apoptosis. Given the involvement of deacetylase in cancer, inhibition or downregulation of HDAC has been investigated as a promising anti-cancer target.
In the study, Kevetrin(TM) potently downregulated HDAC2 in many mutant and null p53 cancer cell lines. HDAC2 is deregulated in many cancers and is emerging as the main deacetylase involved in aberrant pathways of tumor cells in humans. Downregulation of HDAC2 by Kevetrin(TM) strongly induced apoptosis in different types of tumor cell lines.
"We believe we have identified the Mechanism of Action of Kevetrin which explains why Kevetrin(TM) is so effective in a broad spectrum of cancers," says Dr. Krishna Menon, Chief Scientific Officer at Cellceutix. "Downregulation of HDAC2 also explains why Kevetrin(TM) is so effective in drug resistant tumors. Furthermore, since Kevetrin(TM) acts in a non-genotoxic manner and induces potent antitumor activity, we feel that Kevetrin(TM) is a clear standout from all other anticancer drugs. In all my years as a cancer researcher, I haven't seen a drug act like Kevetrin(TM) which is targeting and reacting with virtually every type of cancer."
Cellceutix management confirmed today that its plans are on track for having Kevetrin(TM) manufactured in February 2012 for use in its planned clinical trials as discussed in Cellceutix's press release from December 13, 2011. Cellceutix expects to commence clinical trials at the world's leading cancer center early in 2012.
"In all my years as a cancer researcher, I haven't seen a drug act like Kevetrin(TM) which is targeting and reacting with virtually every type of cancer."
It's very good hook for fund's analysts. May be this small biotech is a future SGEN?
We will see how they will react.
My opinion: it's just a pseudoscience bullshit created to make money. But if funds believe in this company we (small biotech investors) can make some money by playing with them.
Disclosure: I don't have CTIX positions.
01.24.13 by BiotechInvest
The story of biotech scam is continue...
Cellceutix Reports In Vivo Tumor Shrinkage in Renal Cancer
7:00a ET January 24, 2013 (Market Wire)
Cellceutix Corporation (OTCBB: CTIX) (the "Company"), a clinical stage biopharmaceutical company focused on discovering small molecule drugs to treat unmet medical conditions, including drug-resistant cancers and autoimmune diseases, announces today that the Company has received results from in vivo studies conducted at Beth Israel Deaconess Medical Center ("BIDMC"). Cellceutix had earlier reported that they entered into an agreement with BIDMC, a teaching hospital of Harvard Medical School, on an innovative research project with Kevetrin(TM), the Company's flagship anti-cancer drug. BIDMC wishes to exploit the nuclear and/or mitochondrial pro-apoptotic function of p53 in melanoma and renal cell carcinoma, two types of cancer that are particularly resistant to therapy.
Research by BIDMC combined Kevetrin(TM) with sunitinib on cell line 786, a drug-resistant renal cancer. Cellceutix was advised by the researchers that "the Kevetrin/sunitinib combination is the first we've used in which actual tumor shrinkage is noted."
Per protocol, Cellceutix has formally notified BIDMC that it has a strong interest in further collaborative efforts to develop combination treatments with Kevetrin and multikinase VEGF receptor antagonists, particularly VEGFR2 inhibitors. Cellceutix has provided the requested information that will be used to investigate a Specialized Programs of Research Excellence (SPORE) grant for a phase 2 clinical study.
Renal cell carcinoma is the most common type of kidney cancer in adults. BIDMC is the lead recipient of the prestigious National Cancer Institute-funded kidney cancer SPORE grant.
About SPORE In 1992, the National Cancer Institute (NCI) established the Specialized Programs of Research Excellence (SPORE), specialized cancer center grants to promote and speed up the exchange between interdisciplinary research in the laboratory to the treatment of patients in the clinical care setting.
SPORE programs allow laboratory and clinical scientists to work collaboratively to plan, design, and implement research programs with the greatest potential to impact cancer prevention, detection, diagnosis and treatment.
on cell line 786?
Come on...
Cellceutix management confirmed today that they are lairs. But it's possible to use this run up to make some money...
But beware this biotech scam if you want to be long with it. Everybody who said that mutated p53 might be activated is a liar. Mice and rats with hetero KO of p53 gene (one allele is still active) are dying from cancer much sooner that wild type.
So let us make some money with CTIX liars now before this scum will be crashed.
02/25/2013 by BiotechInvest
It's time to buy CTIX to make money with together liars. May it's amoral but moral investors never win in this world.
CTIX management persuaded some big money to buy CTIX stock and persuaded some investors that their drug Kevetrin can resurrect dead "Guardian Angel Gene" i.e. activate even mutated p53 (dead) that lost even DNA binding site.
Well done! Bravo!
Disclosure: I'm in for CTIX crazy run up to $3-5 range. Will buy CTIX today.
03/05/2013 by BiotechInvest
It was too early... Will wait before May
Sure that they still don't know the results of phase I and sent a fake abstract. Now may they already know and start to sell stocks before crash.
Any way these two liars (and 2 employee of CTIX) are already millionaires. There is only one problem: there are millionaires on the paper - when they will start to realize the papers (CTIX stocks) pps will go to 0.something and their millions will evaporate...
"Beware biotech scum but try to make money even on biotech scum"
BiotechInvest
Cellceutix Corporation (OTCBB: CTIX) (the "Company"), a clinical stage biopharmaceutical company focused on discovering small molecule drugs to treat unmet medical conditions, including drug-resistant cancers and autoimmune diseases, today announces that the Company is submitting an abstract for the upcoming 2013 American Society of Clinical Oncology ("ASCO") Annual Meeting to be held in Chicago, Illinois on May 31 to June 4. The presentation is entitled: "A Phase 1 Dose-Escalation, Safety, Pharmacokinetic, Pharmacodynamic Study of Thioureidobutyronitrile (Kevetrin(TM)), a Novel p53 Targeted Therapy in Patients with Advanced Solid Tumors."
Disclosure: I don't have CTIX now but will monitor this stock for possible "in" or short positions.
04/09/2013 by BiotechInvest
An Interview With Leo Ehrlich, Cellceutix CEO
Mar 14 2013, 09:18 | 29 comments
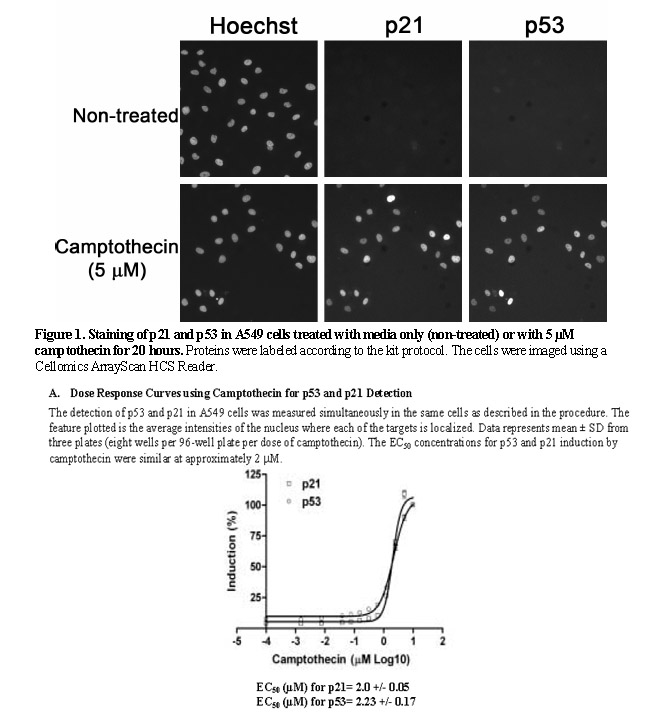
Q: Cellceutix has identified p21 as a biomarker as a barometer of p53 expression for the clinical trial. Seeing activity of p53 without toxic side effects could be the Holy Grail for developing a new cancer treatment. When do you expect results from testing of the p21 biomarker? What are your expectations?
A: We are anticipating the tests to be run in mid-March and the results to follow shortly thereafter. Honestly, we were extremely pleased that the Dana-Farber laboratory is running these tests so early in the trial. It is a "no lose" situation for us as we did not anticipate biomarker testing, nor did we expect to see any activity, at this early stage and low dosing levels. If p21 activity is shown, we think that we have hit a home run, but if activity is not demonstrated, we will not be the least bit disappointed at this juncture in the trial. We will simply then wait for the testing at higher doses as we expected, where we are very optimistic that we will see p21 expression at that time.
Well, I see now that Leo is a good liar so he got abbreviation LL (from Leo Liar).
p21 is real downstream target for p53 so if Kevetrin treatment not increase p21 level the conclusion is simple - it is a fake.
My question is - why these two liars LL and KL never tested their Kevetrin in simple cell-based assay provided by Thermo Scientific? This assay is even shown here with A549 cells. Any lab tech can do this assay for 20 hours (overnight). Cost is several hundred dollars.
And "Kevetrin is effective in mouse models of human lung cancer : A549"
I think that the answer is simple here: Kevetrin will not activate p53 in this model so p21 will not be induced (upregulated). And fake biotech created by two liars will be revealed.
So, we will see how LL and KL will explain the absence of p21 marker in clinical trial. Too low doses? Magic action of Kevetrin that passed p21 upregulation and induce apoptosis in cancer cells directly?
Or something else?
Disclosure: I don't have CTIX shares now, will wait for p21 marker results.
10/02/13 by BiotechInvest
CTIX is in "Strong Sell" status now - two liars LL and KL did another trick - bought some biotech garbage (PYMX assets). Why they did it? The reason is simple: they know that truth about their 2 "blockbuster" drugs Kevetrin and psoriasis drug will be revealed soon so they need something else to attract naive biotech investors. "New antibiotic" is good idea but why the biotech Polymedics that had it filed a bankruptcy? Because nobody was care about this "new and effective antibiotic".
It's funny story about how 2 liars become rich and fool so many investors including one fund ($10M). But it looks like that this story is close to usual end: LL and KL become millionaires and investors lose their money...
03/04/16 By BiotechInvest
I have changed status of CTIX from scam/fraud biotech to "good" biotech with strong development potential. Main reason is CTIX development of PYMX new antibiotic.
My recommendation is BUY and HOLD